- English
- Sociology
- Functional English
- Psychology
- Mathematics
- Philosopy
- Physics
- Computer Science
- Chemistry
- Entrepreneurship
- Biology
- Informatics Practices
- Geography
- Multimedia & Web Technology
- Economics
- Biotechnology
- Business Studies
- Physical Education
- Accountancy
- Fine Arts
- Political Science
- History
- Agriculture
CBSE Guess > Papers > Question Papers > Class XII > 2004 > Chemistry > Delhi Set-III
CHEMISTRY (Set III—Delhi)
Except for the following questions, all the remaining questions have been asked in Set I and Set II.
Q. 1. What makes alkali metal halides sometimes coloured, which are otherwise colourless? 1
Q. 2. State Henry's law about the solubility of a gass in a liquid. 1
Q. 3. Define the order of a reaction. 1
Q. 6. What is meant by entropy driven reaction? How can a reaction with positive changes of enthalpy and entropy be made entropy driven? 2
Q. 7. Give chemical evidence for each of the following: 2
(i) Fluorine is a stronger oxidising agent than chlorine.
(ii) +1 oxidation state is more stable for thallium than that for gallium.
Q. 13. Write the IUPAC name of [Co(en)2Cl2] CI and draw the structures of all the isomers with this formula of complex. 3
Q. 14. Describe the following in brief: 3
(i) Carbon dating
(ii) Thermonuclear reactions
(iii) Group displacement law
Q. 15. How are antiseptics distinguished from disinfectants? Give two examples of each. 3
Q. 25. (a) Assign appropriate reason for each of the following:
(i) Aluminium is highly electropositive yet It is used as a structural metal.
(ii) White phosphorus is much more reactive than red phosphorus.
(iii) Hydrogen fluoride is a much weaker acid than HCI in aqueous solution.
(iv) Sulphur in vapour state exhibits some paramagnetic behaviour.
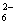 or XeF4 . 5
or XeF4 . 5
| Chemistry 2004 Question Papers Class XII | |||||||
| Delhi | Outside Delhi | Compartment Delhi | Compartment Outside Delhi | ||||
| Set I | Set I | Set I | Set I | ||||
| Set II | Set II | Set II | Set II | ||||
| Set III | Set III | ||||||
