- English
- Sociology
- Functional English
- Psychology
- Mathematics
- Philosopy
- Physics
- Computer Science
- Chemistry
- Entrepreneurship
- Biology
- Informatics Practices
- Geography
- Multimedia & Web Technology
- Economics
- Biotechnology
- Business Studies
- Physical Education
- Accountancy
- Fine Arts
- Political Science
- History
- Agriculture
CBSE Guess > Papers > Question Papers > Class XII > 2004 > Chemistry > Compartment Outside Delhi Set-II
CHEMISTRY (Set II—Compartmet Outside Delhi)
Except for the following questions, all the remaining questions have been asked in Set I.
Q. 1. What happens when ferrimagnetic 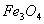 is heated at 850 K and why? 1
is heated at 850 K and why? 1
Q. 2. A and B liquids on mixing produce a warm solution. Which type of deviation from Raoult's law is there? 1
Q. 3. How is acetone obtained from 2-bromopropane? 1
Q. 4. How is phenol obtained from aniline? 1
Q. 6. Justify the following statements: 2
(a) An ezothermic reaction is always thermodynamically spontaneous.
(b) The entropy of a substance increases on going from liquid to vapour state at any temperature.
Q. 8. (a) The standard reduction potential for the Zn2+(aq) I Zn (s) half cell is 0.76 V. Write the reactions occu ring at the electrodes when coupled with standard hydro gen electrode (SHE).
(b) Can a nickel spatula be used to stir a solution of CuSO4 ? Support your answer with reason 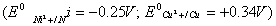 .2
.2
Q. 10. Give reasons:
(a) Xenon does not form fluorides such as XeF3 and XeF5 .
(b) Out of noble gases, only Xenon is known to form real chemical compounds.
Q. 13. Justify the following statements: 3
Q. 23. (a) List two major classes of antibiotics and give one example of each class.
(b) Write one example of each of the following:
(i) Azo dye (ii) Anthraquinone dye 3
Q. 25. Name the chief ores of zinc and mercury. Describe the reactions involved in the extraction of these metals from the respective ores. 5
| Chemistry 2004 Question Papers Class XII | |||||||
| Delhi | Outside Delhi | Compartment Delhi | Compartment Outside Delhi | ||||
| Set I | Set I | Set I | Set I | ||||
| Set II | Set II | Set II | Set II | ||||
| Set III | Set III | ||||||
