- English
- Sociology
- Functional English
- Psychology
- Mathematics
- Philosopy
- Physics
- Computer Science
- Chemistry
- Entrepreneurship
- Biology
- Informatics Practices
- Geography
- Multimedia & Web Technology
- Economics
- Biotechnology
- Business Studies
- Physical Education
- Accountancy
- Fine Arts
- Political Science
- History
- Agriculture
CBSE Guess > Papers > Question Papers > Class XII > 2004 > Chemistry > Compartment Outside Delhi Set-I
CHEMISTRY (Set I—Compartmet Outside Delhi)
Q. 1. What is the utility of piezoelectric crystals? 1
Q. 2. Two liquids X and Y boil at 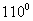 C and 130° C respectively. Which one of them has higher vapour pressure at 50° C? 1
C and 130° C respectively. Which one of them has higher vapour pressure at 50° C? 1
Q. 3. Define activation energy of a reaction. 1
Q. 4. How is ethanoic acid industrially obtained from ethanal? 1
Q. 5. How is methyl nitrile obtained from ethanoyl chloride? 1
Q. 6. On the basis of Heisenberg's uncertainty principle, show that electron can not exist within the atomic nucleus.
(Nuclear radius 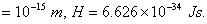 ) 2
) 2
Q. 7. The concept of coupling reactions is useful in making the occurrence of a non-spontaneous reaction possible. Explain giving one suitable example. 2
Q. 8. How would you explain the sharp, increase in the molar conductivity of a weak electrolyte on dilution? How is the molar conductivity related to the degree of dissociation of a sparingly soluble salt? 2
Q. 9. (a) What prompted Bartlet to the discovery of noble gas compounds? 2
(b) State two important uses of noble gases.
Q. 10. Does the presence of two chiral carbon atoms always make the molecule optically active? Explain giving an example. 2
Q. 11. Explain the following giving suitable examples: 2
(a) Kolbe's reaction
(b) Williamson synthesis
Q. 12. How are the following prepared?
(a) Dacron (b) Bakelite
Q. 13. (a) Explain why the uncertainty principle is significant only for the motion of subatomic particles and is negligible for the macroscopic objects.
(b) In what way does the bonding molecular orbital in a molecule of hydrogen differ from its antibonding molecular orbital? 3
Q. 14. (a) Iron ( II ) oxide has a cubic structure and each unit cell has side 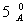 . If the density of the oxide is 4 gm cm>sup>-3 , calculate the number of Fe2+ and O2- ions present in each unit cell. (Molar mass of
. If the density of the oxide is 4 gm cm>sup>-3 , calculate the number of Fe2+ and O2- ions present in each unit cell. (Molar mass of 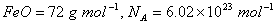 )
)
(b) In a crystal of zinc sulphide, zinc occupies tetrahedral voids. & What is the co ordination number of zinc? 3
Q. 15. 45 g of ethylene glycol C2H6O2 is mixed with 600 g of water. Calculate:
(a) freezing point depression
(b) freezing point of the solution 3
Q. 16. How does the Gibb's energy change when a spontaneous reaction occurs? Show with the help of a diagram how Gibb's energy changes during the course of a reactIon. 3
Q. 17. Silver is electro deposited on a metallic vessel of surface area 800 cm2 by passing current of 0.2 amp for 3 hours Calculate the thickness of silver deposited
(Density of silver =10.47 g cm-3, Atomic mass of silver =107.92 amu) 3
Q. 18. (a) A first order reaction is 15% completed in 20 minutes. How long will it take to complete 60%?
(b) What is the significance of rate constant in a rate law? 3
Q. 19. (a) What are the two types of emulsions and how do they differ from one another? Give one example of each.
(b) Which one of the following electrolytes is most effective for the coagulation of ![]() sot and why?
sot and why?
Or
(a) Which will be absorbed more readily on the surface of charcoal and why? 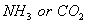 .
.
(b) In what way are multimolecular colloids different from macromolecular colloids? Give one example for each.
(c) State the principle of electrodialysis. 3
Q. 20. (a) A co-ordination compound has the formula 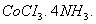 It does not liberate NH but forms a precipitate of AgCl on treatment with
It does not liberate NH but forms a precipitate of AgCl on treatment with 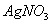 solution Write the structure and IUPAC name of the complex.
solution Write the structure and IUPAC name of the complex.
(b) Name two properties of the central metal ion which enable it to form stable complex entities.
(c) The formation of complex compounds finds application in the extraction of some metals. Furnish one example to support the above statement. 3
Q. 21. (a) How can the course of a reaction be known by using radioisotopes?
(b) Describe the principle of Breeder reactor.
(c) Complete the following nuclear reactions:
(i) 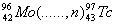
(ii) 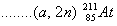 3
3
Q. 22. (a)Give chemical tests to distinguish between the following pairs of compound:
(i) Phenol and Benzoic acid
(ii) Benzaldehyde and Acetophenone
(iii) Give IUPAC name of CH3CONH. C6H5 . 3
Q. 23. Give reasons for the following observations: 3
(a) It is difficult to prepare pure amines by ammonolysis of alkyl halides.
(b) Nitrobenzene does not undergo Friedel Craft's alkylation.
(c) Electrophilic substitution in case of aromatic amines takes place more readily than in benzene.
Q. 24. Define the following and give one example of each: 3
(i) Antihistamines
(ii) Disinfectants
(iii) Antacids
Q. 25. Give reasons for the following facts:
(a) Anhydrous AIcI3 acts as a catalyst.
(c) 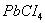 is less stable than
is less stable than 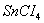
(d) Bond dissociation energy of F2 is less than that of Cl2 .
(e) 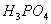 is a diprotic acid. 5
is a diprotic acid. 5
Or
(a) Name the chief ore of aluminium and describe the principle of extraction of aluminium metal from its ore.
(b) How is it that aluminium though an eleètropositive metal, finds extensive use as a structural material? 4, 1
Q. 26. Give reasons:
(a) ![]() is a strong reducing agent whereas
is a strong reducing agent whereas 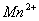 is not. (Cr = 24, Mn = 25)
is not. (Cr = 24, Mn = 25)
(b) The transition metal ions such as
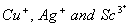 are colourless.
are colourless.
(c) The enthalpies of atomizations of transition metals of 3d series do not fol low a regular trend throughout the series.
(d) The radius of 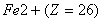 is less than that of Mn2+ (Z = 25).
is less than that of Mn2+ (Z = 25).
(e) Chemistry of the actinoids is much more complicated than that of the lanthanoids. 5
Q. 27. (a) Write down the structures and names of the products formed when D- glucose is treated with
(i) Acetic anhydride
(ii) HI
(iii) Conc. HNO3
(b) What is photosynthesis? Write its importance in the biological world. 3, 2
| Chemistry 2004 Question Papers Class XII | |||||||
| Delhi | Outside Delhi | Compartment Delhi | Compartment Outside Delhi | ||||
| Set I | Set I | Set I | Set I | ||||
| Set II | Set II | Set II | Set II | ||||
| Set III | Set III | ||||||
