CBSE Guess > Papers > Question Papers > Class XII > 2005 > Chemistry > Delhi Set-I
CHEMISTRY 2005 (Set-I Delhi)
Q. 1. What is the maximum possible coordination number of an atom in an hcp crystal structure of an element? 1
Q. 2. State the formula relating pressure of a gas with its mole fraction in a liquid solution in contact with it. 1
Q. 3. Express the relation between the half-life period of a reactant and its initial concentration if the reaction involved is of second order. 1
Q. 4. How are formalin and trioxane related to methanal? 1
Q. 5. Why are primary amines higher boiling than tertiary amines. 1
Q. 6. Show that the Helsenberg Uncertainty Principle is of negligible significance For an object of 10 -6 kg mass. 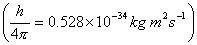
Or
On the basis of Heisenberg Uncertainty Principle show that the electron (mass=9 x 10 -31 kg) cannot exist within an atomic nucleus of radius 10 -15 m.
Q. 7. On the basis of the following 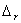 G ° values at 1073 K:
G ° values at 1073 K:
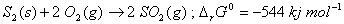
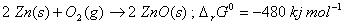
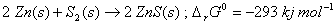
Show that the roasting of zinc sulphide to form zinc oxide is a spontaneous process. 2
Q. 8. Write one chemical reaction each to show that
(a) Tin (II) chloride is a reducing agent.
(b) Chlorine gas can be obtained from bleaching powder. 2
Q. 9. Describe the steps involved in the preparation of either potassium dichro- mate from sodium chromate or potassium permanganate from manganese dioxide. 2
Q. 10. What are enantiomers and diastereomers? Differentiate between chiral and achiral molecules. 2
Q. 11. Give an illustration of Reimer-Tiemann reaction. 2
Q. 12. How is bakelite made and what is its major use? Why is bakelite a thermo-setting polymer' 2
Q. 13. (a) What is meant by linear combination of atomic orbitals?
(b) Illustrate bonding and antibonding molecular orbitals based on bomo-nuclear dihydrogen molecule. 3
Or
What kinds of molecular forces are expected to exist between the species in any three of the following pairs constituting mixtures?
(a) He and N2
(b) Cl2 and NO-3
(c) NH3 and CO2
(d) H2 S and HBr
Q. 14. Aluminium metal forms a cubic close-packed crystal structure. Its atomic radius is 125 x 10 -12 m.
(a) Calculate the length of the side of the unit cell.
(b) How many such unit cells are there in 1.00 m 3 of aluminium? 3
Q. I5. A solution is made by dissolving30 g of a non-volatile solute in 90 g of water. It has a vapour pressure of 2.8 kPa at 298 K. At 298 K, vapour pressure of pure water is 3.64 kPa. Calculate the molar mass of the solute.3
Q. 16. Comment on the validity of the following statements, giving reasons: 3
(a) Thermodynamically an exothermic reaction is sometimes not spontaneous.
(b) The entropy of steam is more than that of water at its boiling point.
(c) The equilibrium constant for a reaction is one or more if 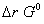 for it is less than zero.
for it is less than zero.
Q. 17. A first order reaction takes 69.3 minutes for 50% completion. Set up an equation for determining the time needed for 80% completion of this reaction. (Calculation of result is not required) 3
Q. 18. Illustrate with examples: 3
(a) Lyophilic and Lyophobic sols
(b) Multimolecular and Macromolecular colloids
(c) Homogeneous and Heterogeneous catalysis.
Q. 19. The E° values in respect of electrodes of chromium (Z = 24), manganese (Z = 25) and iron (Z =26) are:
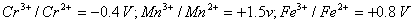
On the basis of the above information compare the feasibilities of further oxidation of their +2 oxidation states.3
Q. 20. Draw a sketch to show the splitting-of d-orbitals in an octahedral crystal field. State for a d 6 ion how the actual configuration of the split d-orbitals in an octa- hedral crystal field is decided by the relative values of 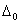 and P. 3
and P. 3
Q. 21. (a) Write the structural formula of 3-phenylprop-2-enal.
(b) Write one chemical equation each to illustrate the following reactions:
(i) Aldol condensation
(ii) Cannizzaro's reaction 3
Q. 22. (a) Assign a reason for each of the following statements:
(i) Alkylamines are stronger bases than arylamines.
(ii) Acetonitrile Is preferred as solvent for carrying out several organic reactions.
(b) How would you convert methylamine into ethylamine? 3
Q. 23. When the nuclides 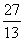 Al,
Al, 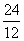 Mg and
Mg and 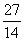 Si are separately subjected to
Si are separately subjected to
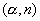 nuclear reactions, three separate new nuclides are produced, each of which further undergoes one positron emission finally giving stable nuclei. Write the nuclear equations for the reactions involved in these cases. 3
nuclear reactions, three separate new nuclides are produced, each of which further undergoes one positron emission finally giving stable nuclei. Write the nuclear equations for the reactions involved in these cases. 3
Q. 24. (a) State the function along with one example each of:
(i) Antihistamines
(ii) Antioxidants
(b) What are hybrid propellants? 3
Q. 25. (a) Define electrical conductivity and molar conductivity of a solution and write the units of molar conductivity.
(b) The E° values corresponding to the following two reduction electrode pro cesses are:
(i) 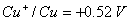
(ii) 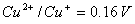
Formulate the galvanic cell for their combination. What will be the standard cell potential for it? Calculate 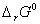 for the cell reaction. (F=96500 C mol -1 ) 2, 3
for the cell reaction. (F=96500 C mol -1 ) 2, 3
Or
(a) In the button cell, widely used in watches and other devices, the following reaction takes place:
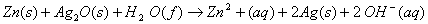
Determine E° and 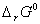 for the reaction.
for the reaction.
Given 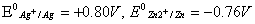
(b) Explain with examples the terms weak and strong electrolytes. How can these be distinguished?
Q. 26. (a) Assign an appropriate reason for each of the following statements:
(i) 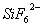 is known but
is known but 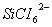 is not known.
is not known.
(ii) More metal fluorides are ionic in nature than metal chlorides.
(iii) Solid phosphorus pentachloride exhibits some ionic character.
(b) Write the structural formulae for the following: 3, 2
(i) BrF3
(ii) XeOF4
Or
(a) Assign a reason for each of the following:
(i) In group 14 the tendency for catenation decreases with increasing atomic numbers.
(ii) In group 15 the bond angle H - M - H decreases in the following order
NH3 (107.8°), PH3 (93.6°), AsH3 (91.8°)
(iii) Sulphur hexafluoride is used as a gaseous electrical insulator.
(b) Complete the following reaction equations: 3, 2
(i) R2 SiCI2 + H2 O
(ii) XeF4 + H2 O
Q. 27. (a) Write chemical equations for the reactions of glucose with
(i) acetic anhydride and
(ii) ammoniacal silver nitrate solution.
(b) Draw simple Fischer projections of D-glucose and L-glucose.
(c) What do you understand by replication by DNA? How does DNA differ from RNA structurally? 2, 1, 2
Or
(a) Write the following about protein synthesis:
(i) Name the location where protein synthesis occurs.
(ii) How do 64 codons code for only 20 amino acids?
(ii) Which of the two bases of the codon are most important for coding?
(b) What deficiency diseases are caused due to lack of vitamins A, B, B6 and K in human diet? 3, 2
- English
- Sociology
- Functional English
- Psychology
- Mathematics
- Philosopy
- Physics
- Computer Science
- Chemistry
- Entrepreneurship
- Biology
- Informatics Practices
- Geography
- Multimedia & Web Technology
- Economics
- Biotechnology
- Business Studies
- Physical Education
- Accountancy
- Fine Arts
- Political Science
- History
- Agriculture
