- English
- Sociology
- Functional English
- Psychology
- Mathematics
- Philosopy
- Physics
- Computer Science
- Chemistry
- Entrepreneurship
- Biology
- Informatics Practices
- Geography
- Multimedia & Web Technology
- Economics
- Biotechnology
- Business Studies
- Physical Education
- Accountancy
- Fine Arts
- Political Science
- History
- Agriculture
CBSE Guess > Papers > Question Papers > Class XII > 2003 > Chemistry > Delhi Set -I
CHEMISTRY—2003 (Set I—Delhi)
Q. 1. Write all the four quantum numbers of the electron in the outermost shell of rubidium (At. no. = 37) atom.1
Q. 2. What type of hybridization is associated with N in NH 3 ? What is the expected bond angle in NH3? 1
Q. 3. Mention one property which is caused due to the presence of F-centre in a solid.
Q. 4. Write Nernst equation for single electrode potential. 1
Q. 5. Complete the nuclear equation: 1
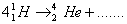
Q. 6. How does chemical adsorption of a gas on a solid vary with temperature? 1
Q. 7. Mention the chief reason for the anomalous behaviour of lithium in Group of the periodic table. 1
Q. 8. Why is the third ionization energy of manganese (At. no. =25) unexpectedly high? 1
Q. 9. Write IUPAC name of the complex Na3 [Cr (OH)2 F4]. 1
Q. 10. Mention two main functions of lipids. 1
Q. 11. Calculate the wavelength of an electron that has been accelerated in a particle accelerator through a potential difference of 100 million volts. 2
(1 eV= 1.6 x 10-19 J, me= 9.1 x 10-31 kg, h= 6.6 x 10-34Js.
Q. 12. Caesiurn chloride crystallises as a body centred cubic lattice and has a density of 4.0 g cm -3 . Calculate the length of the edge of the unit cell of caesium chloride crystal. 2
[Molar mass of CsCI = 168.5 g mol-1 , Na = 6.02 x 1023 mol-1]
Q. 13. What are non-ideal solutions? Explain as to why non-ideal solutions deviate from Raoult's law. 2
Q. 14. The standard free energy change for a reaction is -212.3 kJ mol -1 . If the enthaply of the reaction is - 216.7 kJ mol -1 , calculate the entropy change for the reaction. 2
Q. 15. The rate constant of a reaction is 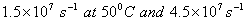 t. Calculate the value of activation energy, E a for the rection.
t. Calculate the value of activation energy, E a for the rection.
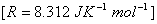
Q. 16. What are photochemical reactions? Explain the machanism of the photo- chemical reaction occurring between hydrogen and chlorine gas. 2
Q. 17. One milligram of 90Sr was absorbed by a new-born child. How much of 90Sr will remain in his bones after 20 years? The half-life of 90Sr is 28.1 years. 2
Q. 18. How are the following sole produced: 2
(a) Sulphur sol
(b) Collodion
Q. 19. Write IUPAC names of the following: 2
(i) 
(ii) CH3 - NH - CH2 -CH - CH3
Q. 20. Write one reaction each to exemplify the following:
(a) Aldol condensation
(b) Friedel-Crafts reaction
Q. 21. How do you account for the following: 2
(a) All scandium salts are white. (At. No. of Sc = 21)
(b) The first ionization energies of the 5d transition elements are higher than those of the 3d and 4d transition elements in respective groups.
Q. 22. Using the valence bond approach, deduce the shape and magnetic character of 2 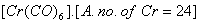
Q. 23. What are elastomers? Write the chemical equation to represent the preparation ration of Buna-S. 2
Q. 24. Name the chemical components which constitute nucleotides. Write any two functions of nucleotides in a cell. 2
Q. 25. What are hormones? State the function of the following hormones: 2
(a) Testosterone
(b) Oxytocin
Q. 26. Give one important use of each of the following: 2
(i) Bithional
(ii) Chloramphenicol
(iii) Streptomycin
(iv) Paracetamol
Q. 27. Explain as to why there is a rise in boiling point when a non-volatile solid is dissolved in a liquid.
0.90 g of a non-electrolyte was dissolved in 87.90 g of benzene. This raised the boiling point of benzene by 0.250 C. If the molecular mass of the non-electrolyte is 103.0 g mol -1 , calculate the molal elevation constant for benzene. 3
Q. 28. State the law of thermodynamics that was first formulated by Nernst in 1906. What is the utility of this law?
The equilibrium constant for the reaction 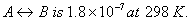 Calculate the value of
Calculate the value of 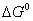 for the reaction.
for the reaction.
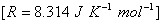
Predict the feasibility of the reaction under standard states. 3
Q. 29. Calculate the cell emf at 25° C for the following cell:
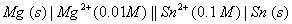

Calculate the maximum work that can be accomplished by the operation of this cell. 3
Q. 30. Write reactions and conditions for the following conversions: 3
(i) Chloroform into diethylcarbonate
(ii) Phenol into salicylic acid
(iii) 2-propanone into 2-inethyl-2-propano
Q. 31. Write chemical tests to distinguish between: 3
(i) Phenol and Benzoic acid
(ii) Propanal and propanone
(iii) Formic acid and Acetic acid
Q. 32. (a) Starting from a sample of chromite ore, how is potassium dichromate prepared? Describe all the steps involved with chemical equations.
(b) Write the balanced chemical equation for the reaction between an acidified solution of potassium dichromate and potassium iodide. 3
Q. 33. (a) How is aniline obtained from benzene?
(b) Why are the secondary amines more basic than primary amines? Explain.
(c) Write the complete chemical reactions for the conversion of aniline to sulphanilic acid.
(d) Mention two important uses of sulphanilic acid.
(e) Write a chemical reaction of aniline which may distinguish it from ethylamine. 5
Q. 34. Explain the following observations: 5
(i) Most of the known noble gas compounds are those of xenon.
(ii) CIF3 exists but FCI3 does not.
(iii) Among the hydrides of elements of Group 16, water shows unusual physical properties.
(iv) Unlike phosphorus, nitrogen shows little tendency for catenation.
(v) Sulphur in vapour state exhibits paramagnetic behaviour.
| Chemistry 2003 Question Papers Class XII | |||||||
| Delhi | Outside Delhi | Compartment Delhi | Compartment Outside Delhi | ||||
| Set I | Set I | Set I | Set I | ||||
| Set II | Set II | Set II | Set II | ||||
