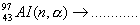CBSE Guess > Papers > Question Papers > Class XII > 2003 > Chemistry > Compartment Delhi Set -II.
CHEMISTRY—2003 (Set II—Compartment Delhi)
Note: Except for the following questions, all the remaining questions have been asked in Set I.
Q. 1. What is the difference between glass and quartz, though both contain SiO 4 units? 1
Q. 7. What is the state of hybridization of nitrogen in the nitrate ion, 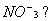 1 1
Q. 8. Name two phenomena that indicate the dull nature of electrons. 1
Q. 10. On mixing equal volumes of water and ethanol, what type of deviation would you expect from Raoult's law? 1
Q. 11. Describe and explain what is observed when 2
(i) a beam of light is passed through a colloidal solution of As 2 S 3 .
(ii) an electric current Is passed through a colloidal solution.
Q. 14. Write the chemical equation representing the system of glyptal. Mention any one of its uses in industry. 2
Q. 26. The specific rate constant for a particular reaction is 2.34 x 10 -3 mol L -1 s -1 at 370 K and 7.50 x 10 -2 mol L -1 s -1 at 400 K. Calculate the activation energy for the reaction.
[R = 8.314 JK -1 mol -1]
Q. 28. (a) Using valence bond theory deduce the geometry and magnetic nature of [Ni (CN) 4 ] 2- .
(At. No. of Ni = 28)
(b) Name the geometrical isomers of [Co(en) 2CI 2 ]+ .
(en = ethylenediamine) 3
Q. 30. (a) Calculate the electrode potential of silver electrode dipped in a 0.1 M solution of silver nitrate at 298 K assuming silver nitrate to be completely dissociated. The standard electrode potential of 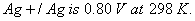
(b) At what concentration of silver ions will this electrode have a potential of zero volt? 3
Q. 31. (a) Complete the following nuclear reactions:
(i) 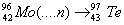 (ii) (ii) 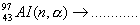
(b) A piece of wood from an old cave gives 6 counts per minute for one gram of carbon. How old is the piece of wood, if in a freshly cut wood the number of counts per minute per gram of carbon is 15.3? 3 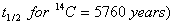
CBSE 2003 Question Papers Class XII
|

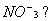 1
1 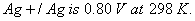
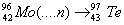 (ii)
(ii)