![]()
CBSE Guess > Papers > Important Questions > Class XI > 2013 > Chemistry By Mr. Sachin Prajapati
CBSE CLASS XI
Chemistry
- Explain why 0-nitrophenol has a lower boiling point than p-nitrophenol?
- Why alkali metals used in photoelectric cells?
- Due to which compound, ozone depletion is caused in Antarctica?
- Which of the element among B, Al, C and Si :-
- Has the highest first ionization enthalpy?
- Has the most negative electron gain enthalpy?
-
- Write the atomic number of the element present in the third period and seventeenth group of the periodic table.
- Out of the elements Cr (Z-24), Mg (Z=12), and Fe (Z=26), identify the element with five electrons in 3d sub shell.
- Critical temperature of ammonia and carbon dioxide are 405.5 K and 304.10 K respectively. Which these gases will liquefy first when you start cooling from 500K to their critical temperature
- An element ‘A’ belongs to group 2 of the periodic table. It shows anomalous behavior from the rest of the elements of its group. It shows a diagonal relationship with another element ‘B’. Chlorides of both ‘A’ and ‘B’ have bridged structure in vapour phase. Identify A and B and draw the structures of their respective chlorides.
- Calculate the mass percent of different elements in sodium sulphate (Na2SO4).
- Why does the rain water normally have a pH of about 5.6? When does it become acid rain?
- The threshold frequency for the ejection of electron from a metal is 5 X 1014 s-1. Will the photon of radiation having energy 3.0 X 10-19 J give photo electric effect or not?
- A metal ‘X’ is present in chlorophyll. Identify the metal ‘X’. write the reaction of this metal with N2.
- An orbital has n=3. What are the possible values of l and m l ?
- Explain the hybridization of PCl5 molecule. Why PCl5 on decomposition gives PCl3 and Cl2 ?
- Consider the chemical reaction:
2SO2 (g) + O2 (g) 2SO3 (g) +189.4 KJ. Indicate the direction in which the equilibrium will shift when:-
2SO3 (g) +189.4 KJ. Indicate the direction in which the equilibrium will shift when:-
- Temperature is increased.
- Pressure is increased.
- Concentration of SO2 is increased.
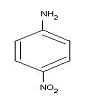
- Write the IUPAC names of the following compound:-
- For the reaction NH4Cl(s) → NH3 (g)+ HCl(g) at 250C, enthalpy change ∇H =+177 kJmol-1and entropy change ∇S = +285JK-1mol-1 .Calculate free energy change ∇G at 250 C and predict whether the reaction is spontaneous or not.
- Explain giving reasons :-
- Boron does not form B3+ ions.
- B-Cl bond has a dipole moment but BCl3 has zero dipole moment.
- Aluminium is used to make transmission cables.
- Alkali metals impart colour to the flame.
- Second ionization enthalpy of Na is higher than Mg.
- Which of the two is bigger in size and why? Cl or Cl-
- What is the conjugate base of HCO3- and H2O ?
- Why are metallic hydrides used for storing hydrogen?
- Name the two methods for estimation of nitrogen.
- Yellow light emitted from a sodium lamp has a wavelength (.) of 580 nm. Calculate frequency (.) and wave number of the yellow light?
- Calculate the oxidation number of Mn in K2MnO4 and N in HNO3.
- Compound ‘A’ with the molecular formula C5H8 reacts with hydrogen in the presence of Lindlar’s catalyst to form a compound ‘B’ with the molecular formula C5H10. ‘A’ on reacting with sodium in liquid ammonia forms a compound ‘C’ with same molecular formula as that of ‘B’. Identify A, B and C. Also write the chemical reactions involved.
- Write the general electronic configuration of p-block and f-block elements.
- Which of the following free radicals is most stable and why? CH2CH3<CH(CH3)2 <C(CH3)3
- Write a short note on the following:-
- Wurtz reaction
- Friedel-crafts alkylation
- Define the following terms:-
- Biological Oxygen Demand (BOD)
- Eutrofication.
- Give reasons for the following:-
- [SiF6]2- is known whereas [SiCl6]2- is not known.
- Diamond is covalent, yet has high melting point.
- BF3 behaves as Lewis acid.
- F has lower electron gain enthalpy than Cl.
- Ionization enthalpy of N is higher than O.
- Falling liquid drops are spherical.
- Conc. HNO3 can be stored in aluminium container.
- Explain structure of Diborane.
Submitted By Mr. Sachin Prajapati
Email: [email protected]
