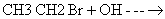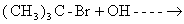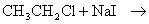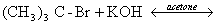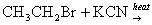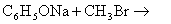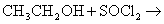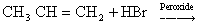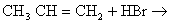![]()
CBSE Guess > Papers > Important Questions > Class XII > 2008 > Chemistry > Chemistry By : Mr. S. Vasudevamurthy
Chemistry
Q. 1. Give one example each of
- Markwonikov’s addition.
- Kharasch effect.
- Sand Meyer reaction
- Diazotisation reaction
- Finkelstein reaction
- Swarts reaction.
- Wurtz reaction
- Wurtz Fittig reaction
- Fittig reaction
- Friediel craft’s acylation reaction of chloro benzene
- Friediel craft’s alkylation reaction.of chloro benzene.
- nitration of chloro benzene.
- sulphonation of chloro benzene.
- Dehydro halogenation
- Zatsev rule.
- chlorination of chloro benzene.
Q. 2. Write the IUPAC names of the following compounds.
- n - butyl chloride
- iso butyl chloride
- secondary butyl chloride
- tertiary butyl chloride.
- ethylidene chloride
- ethylene di chloride
- vinyl chloride.
- p - Cl C6H4 CH2 CH (CH3)2
- m - Cl C6H4 CH2C (CH3)3
- ( CCl3)3 CCl
- o- BrC6H4 CH( CH3) CH2 CH3
- CH3-C (p-Cl C6H4)2/ CH(Br) CH3
Q. 3. Explain the classification of halo alkanes based on
- number of halogen atoms.
- compounds having sp3 C-X bond
- compounds having sp2 C-X bond
- dihalides. Give one example each and their IUPAC names.
Q. 4. Account for the following:
- Halo alkanes have higher boiling point than the corresponding parent alkane.
- Boiling point of halo alkanes RI>RBr>RCl> RF
- Boiling point of 1-Bromo butane >2-Bromo butane> 1-Bromo- 2-methyl propane> 2-Bromo- 2-methyl propane.
- Melting point of p-Dichlo benzene is higher than its ortho and meta isomer.
- Halo alkanes are polar in nature but sparingly soluble in water.
- Iodo alkane can not be prepared by the reaction of alcohol with KI and sulphuric acid. Phosphoric acid is used in place of sulphuric acid.
- Order of reactivity of alcohol with HX is tert alcohol> sec alcohol > primary alcohol..
- Halo arenes can not be prepared by treating phenol with HX or NaX in the presence of sulphuric acid.
- Iodination of benzene is carried out in the presence of HIO3 or HNO3.
- Propane on chlorination gives 2-chloro propane as a major product and not 1-chloro propane.
- Kharasch effect is possible only with HBr and not with HCl and HI.
- Alcohol reacts with thionyl chloride to give pure halo alkane.
- Finkelstein reaction of halo alkane is carried out in the presence of dry acetone.
- Order of reactivity of halo alkanes as per substitution bimolecular nucleophilic is primary halide > secondary halide>tertiary halide.
- Order of reaction as per substitution unimolecular is tertiary halide>secondary halide >primary halide.
- Benzylic halides and allylic halides are more reactive towards nucleophile than halo alkanes.
- Chloro ethene is less reactive towards nucleophile than chloro ethane.
- Halo arenes are less reactive towards nucleophile than halo alkanes.
- SN1 mechanism is ruled out in the reaction of halo arenes with nucleophile.
- Electron with drawing groups like NO2 at ortho and para position with respect to halogen facilitates nucleophillic substitution reaction.
- Electron with drawing groups like NO2 at meta position with respect to halogen has no effect on nucleophillic substitution reaction.
- Halo arenes are less reactive towards electrophile than benzene.
- Although chlorine atom has electron with drawing effect electrophillic substitution occur at ortho and para position.
- Order of reactivity of alkyl halide RI > RBr > RCl > RF
- Halo alkanes react with KCN to give alkyl cyanide as a major product while it gives alkyl isocyanide as a major product with AgCN.
- Halo alkanes give nitrito alkane with KNO2 while nitro alkane with AgNO2.
Q. 5. Explain the following with suitable examples:
- chiral and chirality
- enantiomers
- racemic mixture
- retention of configuration
- inversion of configuration.
Q. 6. Mention the differences between SN1 and SN2 mechanism of halo alkane.
Q. 7. Explain the mechanisms of the following reactions :
Q. 8. Carry out the following conversions :
- Propene to a) Propan-1-ol b) Propan-2-ol
- Ethanol to but-1-yne
- 1-Bromo propane to 2-Bromo propane and vice versa.
- Toluene to benzyl alcohol.
- Benzene to a) 4-bromonitro benzene b) 3-bromonitro benzene.
- Benzyl alcohol to 2-phenyl ethanoic acid.
- Ethanol to a) Propane nitrle b) Ethyl isocyanide.
- Aniline a) Chloro benzene b) Bromo benzene c) Iodo benzene.
- 2-Chloro butane to 3,4- dimethylhexane.
- 2-Methyl-1-propene to 2-chloro-2-methylpropane.
- Ethyl chloride to propanoic acid.
- But-1-ene to n-butyl iodide.
- 2-chloropropane to propan-1-ol
- Isopropyl alcohol to iodoform.
- Chloro benzene to a) p-nitro phenol b) p-chloronitro benzene c) p-chloro
methyl benzene. d) p- chloro acetophenone. e ) p-chloro benzene sulphonic
acid f) 1,4-Dichloro benzene. g ) biphenyl. - Chloroethane to butane.
- tert-butyl bromide to isobutyl bromide.
- Aniline to phenylisocyanide.
- Propene to a) 2,3-dimethyl butane b) n-hexane.
- Tert-butyl bromide to 2-methyl prop-1-ene.
Q. 9. What happens when
- n-butyl chloride is treated with alcoholic KOH.
- bromobenzene is treated with Mg in the presence of dry ether.
- chlorobenzene is subjected to hydrolysis.
- ethyl chloride is treated with aqueous KOH.
- methyl bromide is treated with Na in the presence of dry ether.
- methyl chloride is treated with a)KCN b)AgCN c)KNO2 d)AgNO2
Q. 10. Write the structure of the major organic product in each of the following reactions:
Q. 11. Arrange the compounds of each set in order of decreasing reactivity towards a) SN2 displacement. b) SN1 displacement.
- 2-bromo-2-methylbutane, 1-bromopentane, 2-bromo pentane
- 1-bromo-3-methylbutane, 2-bromo-2-methylbutane,3-bromo-2-methylbutane
- 1-bromo butane, 1-bromo-2-methyl propane, 1-bromo-2-phenyl propane.
Q. 12. Primary halide A(C4H9Br) with alcoholic KOH gives a compound B. B on treatment with HBr gives C which is an isomer of A. A on treatment with Na in dry ether gives a compound D which is different from when n-butyl bromide is reacted with Na in dry ether. Give the structural formula of A. Write the equations of the reactions involved.
Q. 13. Distinguish chemically between a) CH3Cl, CH3Br, CHM3I b) Chloro benzene and chloro methane c) chloro benzene and benzyl chloride d) CHCl3 and CCl4.